An international research team has characterised, in the brain of foetuses, the cell populations responsible for the formation of blood vessels in the prenatal human brain. They are groups of endothelial and mural cells that line the cerebral vasculature in the second trimester of pregnancy. Led by the University of San Francisco, the work has the participation of the Cavanilles Institute of the University of Valencia-CIBERNED, as well as the collaboration of the Health Research Institute (IIS) and the La Fe Hospital in Valencia.
Interactions between neurogenesis and angiogenesis – the process of forming new blood vessels – are known to regulate embryonic brain development. However, there are doubts about the growth processes and maturation stages of vascular cells, especially in the prenatal human brain. The international research team in which the University of Valencia participates has just contributed new data to the study of this unknown vascular maturation, by identifying the cells that promote angiogenesis and that, therefore, would have an important role in the generation of neurons and in the development of the human brain.
As described in the article published in the journal Cell (IF 66,850), among the sets of endothelial cells – which line the interior of the blood vessels – and mural cells – which surround the endothelial cells –, the so-called ‘tracer cells’ predominate, a subtype of endothelial cells that, due to their quantity and location in the middle of the zone of active neurogenesis – the ventricular zone –, could be responsible for coordinating the function of neuronal stem cells, which helps neurogenesis. These tracking cells were morphologically characterised by the professor of Cell Biology at the University of Valencia and co-author of the work José Manuel García-Verdugo, using electron microscopy techniques.
Among the sets of endothelial cells – which line the interior of the blood vessels – and mural cells – which surround the endothelial cells –, the ‘tracer cells’ predominate, a subtype of endothelial cells that, due to their quantity and location in the middle of the zone of active neurogenesis – the ventricular zone –, could be responsible for coordinating the function of neuronal stem cells, which helps neurogenesis
The study analyses vascular development in foetal stages, at spatial and temporal levels, comparing a highly neurogenic area – the Medial Ganglion Eminence – with the cerebral cortex, with less neuronal proliferation. “The vessels in the Eminence form an extensive mosaic-like network, with a high proliferative rate, which coincides with the area responsible for neurogenesis in this region. On the contrary, the vessels of the cortex are distributed radially and show less proliferation”, comments Susana González, researcher at CIBERNED and the Cavanilles Institute of Biodiversity and Evolutionary Biology (ICBiBE) of the University of Valencia and one of the authors of the article.
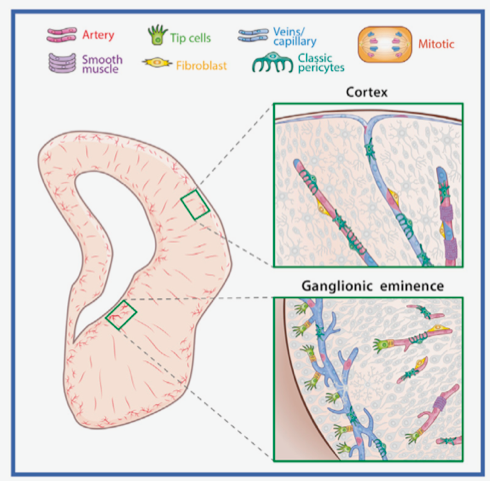 Various endothelial and mural cells contribute to the development of blood vessels in the cerebral cortex and ganglionic eminence of the prenatal human brain.
Various endothelial and mural cells contribute to the development of blood vessels in the cerebral cortex and ganglionic eminence of the prenatal human brain.
The study also suggests that it is at the end of the second trimester of pregnancy when the blood filling of the vessels in this region begins. And it shows that vascular cells in the brain at this stage follow different developmental trajectories and use signaling mechanisms such as collagen, laminin and midkinin to facilitate communication and maturation of the vessels. “The results reveal important mechanisms of vascular maturation”, comments García-Verdugo, “and the fact that neurogenesis and angiogenesis share the same type of signalling suggests that vascular maturation itself is functionally supporting the development of human neuronal progenitors”, concludes the scientist from the University of Valencia.
“The results reveal important mechanisms of vascular maturation, and the fact that neurogenesis and angiogenesis share the same type of signalling suggests that vascular maturation itself is functionally supporting the development of human neuronal progenitors”, José Manuel García-Verdú, professor of Cell Biology at the University of Valencia
“Defects in the adult vasculature are increasingly related to neurological conditions, including Alzheimer’s disease or cerebrovascular arteriovenous malformations”, says Jaime Ferrer Lozano, pathologist at the La Fe University and Polytechnic Hospital of Valencia. “The monitoring of the specific properties of ventral angiogenesis in this study could inform the risk of bleeding in preterm infants. This can happen mainly during the first 72 hours of life, which greatly conditions the evolution of cognitive and sensory development in the medium and long term”, concludes Máximo Vento, Head of the Perinatology Research Group and emeritus researcher at the IIS.
ARTICLE REFERENCE
Ensembles of endothelial and mural cells promote angiogenesis in prenatal human brain. Elizabeth E. Crouch, Aparna Bhaduri, Madeline G. Andrews, Arantxa Cebrian-Silla, Loukas N. Diafos, Janeth Ochoa Birrueta, Kaylee Wedderburn-Pugh, Edward J. Valenzuela, Neal K. Bennett, Ugomma C. Eze, Carmen Sandoval-Espinosa, Jiapei Chen, Cristina Mora, Jayden M. Ross, Clare E. Howard, Susana Gonzalez-Granero, Jaime Ferrer Lozano, Máximo Vento, Maximilian Haeussler, Mercedes F. Paredes, Ken Nakamura, Jose Manuel García-Verdugo, Arturo Álvarez-Buylla, Arnold R. Kriegstein, and Eric J. Huang. CELL. DOI: https://doi.org/10.1016/j.cell.2022.09.004


